121 S. Wilke Road, Suite 301, Arlington Heights, IL 60005
Home and Hospital Visits for Your Convenience
Serving Clients Across 7 Illinois Locations
Recent Blog Posts
How Do I Know If I Can File a Slip-and-Fall Claim?
 You may know that if you are injured by slipping or tripping and falling on someone else’s property, you might be entitled to compensation through a premises liability claim. However, you need to prove various facts for your claim to be successful. Slip and fall cases can be tricky, so it is best to work with one of our experienced Schaumburg, IL slip-and-fall attorneys if you hope to receive as much as you deserve.
You may know that if you are injured by slipping or tripping and falling on someone else’s property, you might be entitled to compensation through a premises liability claim. However, you need to prove various facts for your claim to be successful. Slip and fall cases can be tricky, so it is best to work with one of our experienced Schaumburg, IL slip-and-fall attorneys if you hope to receive as much as you deserve.
What Is the Illinois Premises Liability Act?
Illinois law requires property owners to use reasonable care to maintain safe premises for invitees and licensees. Invitees are those who are on a property for business purposes, such as a delivery person, a retail shopper, a restaurant diner, or a contractor working on a property. Licensees are people who are on a property for social or non-business-related purposes. Both groups are welcome on the property, unlike trespassers.
Are Wrongful Death Claims Only for Car Accidents?
 In 2023, traffic crashes killed more than three people every day in Illinois, for a total of 1,240 fatalities for the year. While wrongful death claims often stem from car, motorcycle, and large truck crashes, other kinds of fatal incidents can also qualify as grounds for eligible survivors to pursue compensation.
In 2023, traffic crashes killed more than three people every day in Illinois, for a total of 1,240 fatalities for the year. While wrongful death claims often stem from car, motorcycle, and large truck crashes, other kinds of fatal incidents can also qualify as grounds for eligible survivors to pursue compensation.
Several factors need to be considered to see if you qualify to bring wrongful death and survival actions in Illinois. Our knowledgeable Palatine wrongful death attorneys can tell you if you are eligible and help you seek maximum compensation.
How Do I Know If I Can File a Wrongful Death Claim?
Most wrongful death claims are based on proving that someone else’s negligence directly caused the death. In this legal context, negligence refers to careless behavior that endangers others. Negligence in car crashes, for example, could be speeding, following too closely, or distracted or drunk driving. Intentional acts, such as assaults, and defective or dangerous product liability could also be grounds to bring a wrongful death action.
What Compensation Can I Get for My Child’s Birth Injuries?
 Having a baby should be a joyous occasion. You plan for months, getting the baby’s room ready, picking a name, and preparing your other children for a new sibling. When your infant is harmed by preventable birth injuries, your entire world can shift. Caring for a child with permanent conditions like cerebral palsy can be very expensive, not to mention the emotional and mental toll it can take on you.
Having a baby should be a joyous occasion. You plan for months, getting the baby’s room ready, picking a name, and preparing your other children for a new sibling. When your infant is harmed by preventable birth injuries, your entire world can shift. Caring for a child with permanent conditions like cerebral palsy can be very expensive, not to mention the emotional and mental toll it can take on you.
If medical negligence caused your infant’s birth injuries, you could be entitled to compensation. However, you need strong evidence proving that negligence was responsible. Medical malpractice cases are complex, and many people wrongly believe that medical professionals are above reproach. You need an experienced Rolling Meadows, IL birth injuries lawyer as your advocate if you hope to secure the full compensation you deserve.
Does Scott’s Law Only Apply to State Troopers?
 On April 7th, 2025, an Illinois State Trooper was seated in his car at the scene of an accident on I-94 in Chicago. The trooper’s squad car was in the right-hand lane with its flashing lights activated. A Chevy Impala hit the squad car, injuring the trooper. His injuries were not life-threatening, but he was transported to a nearby hospital. This collision was the fifth accident caused by a violation of Scott’s Law in 2025. In 2024, one ISP trooper was killed and 12 were injured in Scott’s Law crashes.
On April 7th, 2025, an Illinois State Trooper was seated in his car at the scene of an accident on I-94 in Chicago. The trooper’s squad car was in the right-hand lane with its flashing lights activated. A Chevy Impala hit the squad car, injuring the trooper. His injuries were not life-threatening, but he was transported to a nearby hospital. This collision was the fifth accident caused by a violation of Scott’s Law in 2025. In 2024, one ISP trooper was killed and 12 were injured in Scott’s Law crashes.
It seems that the people most affected by Scott’s Law violations are state troopers. However, this law’s requirements also apply to other kinds of vehicles. Whether inside their vehicles or outside them, working as pedestrians, troopers are not the only ones who can be harmed. Pedestrians often sustain catastrophic or fatal injuries in motor vehicle collisions. If you are injured in a car accident as a driver, passenger, or pedestrian, our experienced Arlington Heights, IL personal injury attorneys will advocate for your interests and fight to maximize your settlement.
Are Dog Attacks Worse for Children?
 Did you know that Chicago’s dog ownership density is the highest in the U.S.? Almost half of Illinois homes own pets, with 32.4 percent of those pets being dogs. Unfortunately, Illinois also ranks high for the annual number of dog attacks and bites. Even worse, the victims are often children. Along with the physical injuries from these attacks, victims often experience severe psychological trauma.
Did you know that Chicago’s dog ownership density is the highest in the U.S.? Almost half of Illinois homes own pets, with 32.4 percent of those pets being dogs. Unfortunately, Illinois also ranks high for the annual number of dog attacks and bites. Even worse, the victims are often children. Along with the physical injuries from these attacks, victims often experience severe psychological trauma.
Under the Illinois Animal Control Act, you may be able to pursue a compensation claim against a dog’s owner or other responsible party for the harm suffered in an unprovoked attack. A dedicated Rolling Meadows, IL dog attack attorney can help you pursue your claim and collect as much compensation as you deserve.
Are Bicyclists Considered Pedestrians in Illinois?
 With spring making its appearance, many people in Illinois are riding their bicycles. The question of whether bike riders are considered pedestrians is common. Because bicycles can travel on both sidewalks and roads, the answer is, "It depends on where the bike is being ridden."
With spring making its appearance, many people in Illinois are riding their bicycles. The question of whether bike riders are considered pedestrians is common. Because bicycles can travel on both sidewalks and roads, the answer is, "It depends on where the bike is being ridden."
While biking can be a fun way to exercise or an inexpensive mode of travel, bicyclists are extremely vulnerable if they are involved in an accident. So are pedestrians. Motorists, pedestrians, and bicyclists need to follow traffic rules to avoid collisions. If you are injured in an accident caused by someone else, one of our skilled Arlington Heights, IL personal injury lawyers can help you get the compensation you deserve.
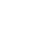
Can I Sue for a Healthcare Acquired Infection?
 People visit doctor’s offices, clinics, hospitals, and other healthcare providers for a variety of reasons. Some patients are sick, seeking treatment for their ailments. Others may have broken a leg, need surgery, or be having a baby. When you are released from these facilities, you expect to be in the same or better condition as you were when you entered. Unfortunately, some patients get more than they bargained for.
People visit doctor’s offices, clinics, hospitals, and other healthcare providers for a variety of reasons. Some patients are sick, seeking treatment for their ailments. Others may have broken a leg, need surgery, or be having a baby. When you are released from these facilities, you expect to be in the same or better condition as you were when you entered. Unfortunately, some patients get more than they bargained for.
Healthcare- or hospital-acquired infections (HCAIs) affect approximately 1.7 million hospital patients yearly, with over 98,000 patients dying as a result. HCAIs are among the top 10 causes of death in the U.S. and are the most common hospital care complications. Patients can acquire infections in other healthcare settings, as well. If you suffered harm from an HCAI or a loved one died, a dedicated Schaumburg, IL medical malpractice lawyer can help you get the compensation you deserve.
Are Hit and Run Accidents Covered by Insurance?
 Illinois law requires drivers to remain at the scene of a car accident that causes injuries or fatalities. Although illegal, some drivers leave accident scenes without stopping to assist injured victims or waiting for police officers to arrive. When you know who caused a car accident, you can often file an insurance claim against the at-fault party’s liability insurance policy. When the driver flees, victims wonder whether they can pursue compensation.
Illinois law requires drivers to remain at the scene of a car accident that causes injuries or fatalities. Although illegal, some drivers leave accident scenes without stopping to assist injured victims or waiting for police officers to arrive. When you know who caused a car accident, you can often file an insurance claim against the at-fault party’s liability insurance policy. When the driver flees, victims wonder whether they can pursue compensation.
Like so many other legal questions, the answer is, "It depends." Whether you are an injured victim or need to file a wrongful death claim after a hit-and-run took your loved one, a knowledgeable Palatine, IL personal injury lawyer can help.
Is a Personal Injury Insurance Claim the Same as a Lawsuit?
 You are probably aware that you could be entitled to compensation if you are injured in an accident caused by someone else. However, you may not understand how the personal injury process works, and that could lead you to make costly mistakes. While most of these cases begin with the victim filing an insurance claim, the insurance company is highly unlikely to pay a fair amount voluntarily. Sometimes, a civil lawsuit and trial are needed to force an insurer to pay as much as it should. For optimal results, you need an experienced Arlington Heights, IL personal injury attorney advocating for your best interests.
You are probably aware that you could be entitled to compensation if you are injured in an accident caused by someone else. However, you may not understand how the personal injury process works, and that could lead you to make costly mistakes. While most of these cases begin with the victim filing an insurance claim, the insurance company is highly unlikely to pay a fair amount voluntarily. Sometimes, a civil lawsuit and trial are needed to force an insurer to pay as much as it should. For optimal results, you need an experienced Arlington Heights, IL personal injury attorney advocating for your best interests.
How Do I Know If I Have a Personal Injury Case?
Most personal injury claims require you to prove that someone else’s intentional acts or negligence caused your accident and injuries. If a defective product caused you harm, product liability claims may be based on strict liability. Many kinds of incidents could provide the grounds you need to file a claim, including:
Can Pre-Existing Conditions Affect My Personal Injury Case?
 Few people go through life without experiencing some kind of injury. When someone is injured in an accident, the new injuries can worsen pre-existing conditions. While the victim may still qualify to collect compensation through a personal injury claim, the liable insurer will probably try to deny liability due to the old injury.
Few people go through life without experiencing some kind of injury. When someone is injured in an accident, the new injuries can worsen pre-existing conditions. While the victim may still qualify to collect compensation through a personal injury claim, the liable insurer will probably try to deny liability due to the old injury.
This type of case can be challenging to deal with alone. You can trust our knowledgeable Rolling Meadows, IL personal injury attorneys to handle your case effectively, always focused on protecting your best interests.
What Kinds of Accidents Can Worsen Pre-Existing Conditions?
You may have an old back injury that, although healed, still flares up occasionally. You might have broken a bone a few years ago that still aches sometimes. You could be prone to seizures due to a concussion in a skiing accident last winter. Old injuries can rear their heads again if you are involved in an accident, such as a:

 Spanish
Spanish